
CMRR
Center for Magnetic Resonance Research, Department of Radiology
Research Highlights
You are here
The ability to acquire whole brain images rapidly (i.e. with short TR) using methods developed in this BTRC has proven to be critically important for detection of resting state networks (RSNs) from resting state functional imaging (R-fMRI) data [1]. Regions of the brain within an RSN display spontaneous fluctuations that are coherent within a network and distinct across different networks [2-6]. RSNs are thought to represent functional connectivity; as such, improved detection of RSNs are a primary target in the Human Connectome Project (http://humanconnectome.org/).
“Resting state” in resting state fMRI does not refer to the brain being at rest, which is never the case; rather it refers to the subject being at “rest” in the scanner, not performing a directed task or exposed to an external stimulus as in normal fMRI.
RSNs have been shown to be related to age and gender [7-9], and particularly the default mode network (DMN) has opened the possibility of using RSNs in clinical applications [10-14].
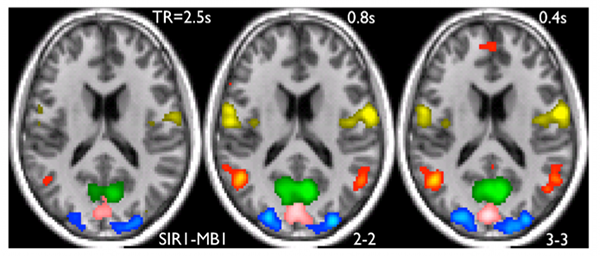
Example of five RSNs are shown in the figure above; three visual area networks (pink/blue/green), the default mode network (red) and a sensori-motor network. The data were acquired using methods we developed recently to increase the speed of whole brain imaging [1]. The data demonstrate that detection of the networks improve with increased speed of acquisition (i.e. decreasing TR). When all possible networks detected in a 100 component ICA analysis were considered, in each case there was improved statistical significance for the detection of these networks for the faster (i.e. short TR ) acquisition.
REFERENCES
1. Feinberg, D.A., S. Moeller, S.M. Smith, E. Auerbach, S. Ramanna, M.F. Glasser, K.L. Miller, K. Ugurbil, and E. Yacoub: Multiplexed echo planar imaging for sub-second whole brain FMRI and fast diffusion imaging. PLoS ONE, 2010. 5(12): p. e15710.
2. Biswal, B., F.Z. Yetkin, V.M. Haughton, and J.S. Hyde: Functional connectivity in the motor cortex of resting human brain using echo-planar MRI. Magnetic Resonance in Medicine, 1995. 34(4): p. 537-41.
3. Lowe, M.J., B.J. Mock, and J.A. Sorenson: Functional connectivity in single and multislice echoplanar imaging using resting-state fluctuations. Neuroimage, 1998. 7(2): p. 119-32.
4. Beckmann, C.F., M. DeLuca, J.T. Devlin, and S.M. Smith: Investigations into resting-state connectivity using independent component analysis. Philos Trans R Soc Lond B Biol Sci, 2005. 360(1457): p. 1001-13.
5. Damoiseaux, J.S., S.A. Rombouts, F. Barkhof, P. Scheltens, C.J. Stam, S.M. Smith, and C.F. Beckmann: Consistent resting-state networks across healthy subjects. Proceedings of the National Academy of Sciences of the United States of America, 2006. 103(37): p. 13848-53.
6. Nir, Y., U. Hasson, I. Levy, Y. Yeshurun, and R. Malach: Widespread functional connectivity and fMRI fluctuations in human visual cortex in the absence of visual stimulation. Neuroimage, 2006. 30(4): p. 1313-24.
7. Esposito, F., A. Aragri, I. Pesaresi, S. Cirillo, G. Tedeschi, E. Marciano, R. Goebel, and F. Di Salle, Independent component model of the default-mode brain function: combining individual-level and population-level analyses in resting-state fMRI, in Magnetic Resonance Imaging. 2008. p. 905-13.
8. Esposito, F., A. Aragri, I. Pesaresi, S. Cirillo, G. Tedeschi, E. Marciano, R. Goebel, and F. Di Salle: Independent component model of the default-mode brain function: combining individual-level and population-level analyses in resting-state fMRI. Magnetic Resonance Imaging, 2008. 26(7): p. 905-13.
9. Biswal, B.B., M. Mennes, et al., Toward discovery science of human brain function, in Proc Natl Acad Sci USA. 2010. p. 4734-9.
10. Greicius, M.D., G. Srivastava, A.L. Reiss, and V. Menon, Default-mode network activity distinguishes Alzheimer's disease from healthy aging: evidence from functional MRI, in Proc Natl Acad Sci USA. 2004. p. 4637-42.
11. Buckner, R.L., A.Z. Snyder, et al., Molecular, structural, and functional characterization of Alzheimer's disease: evidence for a relationship between default activity, amyloid, and memory, in J Neurosci. 2005. p. 7709-17.
12. Garrity, A.G., G.D. Pearlson, K. McKiernan, D. Lloyd, K.A. Kiehl, and V.D. Calhoun, Aberrant "default mode" functional connectivity in schizophrenia, in Am J Psychiatry. 2007. p. 450-7.
13. Sperling, R.A., P.S. Laviolette, et al., Amyloid deposition is associated with impaired default network function in older persons without dementia, in Neuron. 2009. p. 178-88.
14. Fox, M.D. and M.D. Greicius, Clinical applications of resting state functional connectivity, in Front Syst Neurosci. 2010. p. 19.