
CMRR
Center for Magnetic Resonance Research, Department of Radiology
Research Highlights
You are here
Diabetes
Effects of Diabetes on Brain Structure and Function
Diabetes mellitus is a complex metabolic disease that affects many, if not all, of the organs in the body. Whether diabetes has an effect on the central nervous system was uncertain for many years, but as patients with diabetes live longer and better with the disease, the consequences of diabetes on the brain are now becoming apparent. Both hyperglycemia and the hypoglycemia associated with diabetes treatment have been hypothesized to alter brain structure and function. CMRR researchers collaborate with colleagues from the Department of Medicine, Division of Endocrinology and Diabetes to determine the effects of diabetes and its treatments on brain metabolism, structure and function in patients.
HYPOGLYCEMIA UNAWARENESS
More than 25 million Americans have diabetes and hypoglycemia is a common event in the lives of patients who receive insulin as therapy for their disease. Inconvenient and disruptive to daily activities, it can also lead to loss of consciousness, seizures, and death. Patients who experience recurrent episodes of hypoglycemia often develop hypoglycemia unawareness a clinical syndrome in which the first symptom of hypoglycemia is confusion or loss of consciousness. The fear of hypoglycemia unawareness prevents many patients with diabetes from using enough insulin to control their blood sugar, which increases their risk of developing serious side effects. To develop better ways to prevent hypoglycemia unawareness first requires an understanding of the causes of this syndrome. Using high field MR technology CMRR researchers have been testing hypotheses that may explain how the brain adapts to recurrent episodes of hypoglycemia.
GLUCOSE TRANSPORT AND METABOLISM IN THE BRAIN
Glucose is the main fuel for brain function. One way the brain may adapt to hypoglycemia is to have more glucose available to fuel brain activity. The brain may accomplish this either by increasing glucose transport from the blood, decreasing glucose utilization or increasing its storage in the form of glycogen, the main sugar-storing molecule in the brain. Studies at CMRR provided evidence for elevated free glucose levels in the brains of patients with type 1 diabetes and hypoglycemia unawareness [1], but no difference in the rate that the brain uses glucose [2]. CMRR researchers recently developed methods to measure glucose transport and metabolism simultaneously, which will facilitate future studies in this area [3]. They also discovered that after healthy volunteers have their blood sugar levels artificially reduced, their brains made glycogen faster than before (Figure 1), suggesting that patients with hypoglycemia unawareness may have access to higher levels of stored glucose [4]. Current studies investigate if they indeed have higher glycogen levels than controls to shed more light into the mechanism of hypoglycemia unawareness.
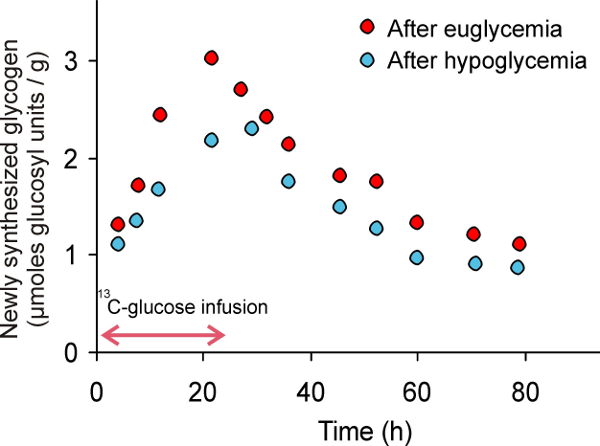
Figure 1. Studies with 13C magnetic resonance spectroscopy show that the brain synthesizes more glycogen after hypoglycemia than after euglycemia.
EFFECTS OF DIABETES ON BRAIN FUNCTION AND STRUCTURE
Patients with diabetes have an increased risk of developing dementia and experiencing a stroke, two brain diseases that can alter brain function and structure. It is unknown if diabetes has an impact on the brain of seemingly normal individuals with long standing diabetes. CMRR researchers utilized a technique called diffusion tensor imaging that measures the magnitude and directionality of water diffusion to assess brain tissue injury in diabetes before it has progressed to the point of detection by more conventional imaging techniques. They discovered that adults with more than 15 years of type 1 diabetes have changes in the microstructure of white matter (Figure 2) that correlate with reduced performance on some cognitive tests [5]. These white matter changes also were functionally connected to areas of the brain with reduced gray matter density [6]. Future studies will determine if these changes are predictive of dementia.
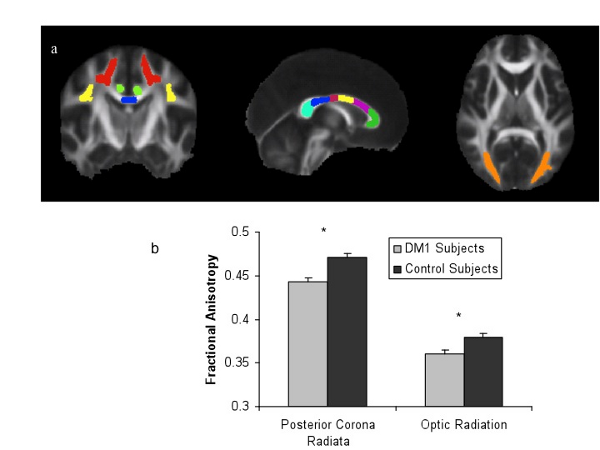
Figure 2. White matter structural integrity is lower in patients with longstanding type 1 diabetes (DM1) relative to healthy controls.
REFERENCES
[1] Criego AB, Tkac I, Kumar A, Thomas W, Gruetter R, Seaquist ER. Brain glucose concentrations in patients with type 1 diabetes and hypoglycemia unawareness. J Neurosci Res 2005;79(1-2):42-47.
[2] Henry PG, Criego AB, Kumar A, Seaquist ER. Measurement of cerebral oxidative glucose consumption in patients with type 1 diabetes mellitus and hypoglycemia unawareness using 13C nuclear magnetic resonance spectroscopy. Metabolism 2010;59(1):100-106.
[3] Shestov AA, Emir UE, Kumar A, Henry PG, Seaquist ER, Öz G. Simultaneous measurement of glucose transport and utilization in the human brain. Am J Physiol Endocrinol Metab 2011; DOI:10.1152/ajpendo.00110.2011.
[4] Öz G, Kumar A, Rao JP, Kodl CT, Chow L, Eberly LE, Seaquist ER. Human brain glycogen metabolism during and after hypoglycemia. Diabetes 2009;58(9):1978-1985.
[5] Kodl CT, Franc DT, Rao JP, Anderson FS, Thomas W, Mueller BA, Lim KO, Seaquist ER. Diffusion tensor imaging identifies deficits in white matter microstructure in subjects with type 1 diabetes that correlate with reduced neurocognitive function. Diabetes 2008;57(11):3083-3089.
[6] Franc DT, Kodl CT, Mueller BA, Muetzel RL, Lim KO, Seaquist ER. High connectivity between reduced cortical thickness and disrupted white matter tracts in long-standing type 1 diabetes. Diabetes 2011;60(1):315-319.